
The compound NaCl is composed of two elements, sodium (Na) and Chlorine (Cl) and together when they react chemically, they form NaCl as the product. Let us take the example of the molar mass of NaCl. 
Compounds are basically substances that are formed when two or more elements react chemically while elements, on the other hand, are naturally occurring substances found mostly in abundance on Earth or in the atmosphere. That is, the molar mass of a substance= 1 gram/I moleĬalculating the molar mass of NaCl can be extremely simple only if you know the difference between elements and compound and have a knowledge about the molecular weight of the respective elements. The molar mass of a compound is also known as its molecular weight and is the total mass in grams of the atoms that form one mole of a certain molecule. Once we are aware of the moles required for a particular experiment, we can easily use the concept and notion of molar mass for determining the grams of substance required for our purpose. A mole is nothing, but a unit used for measurement which helps in comparing particles and mass of a given compound. After various calculations and observations, a scientist discovers the number of moles required for a particular experiment based on which we calculate the molar mass. For all laboratory calculations, a tool called the analytical balance is used to measure the weights in grams. Thus, a change in the molecular mass of a compound can greatly vary the results of any scientific calculations. It becomes essential that we measure these molecules in all the experiments to get accurate results for our products in the experiment. We know that all things around us are occupied by matter whose building block is molecules. 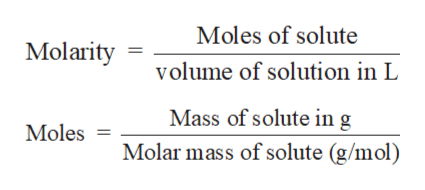
Before we begin to calculate the molar mass of NaCl or any other compound for that matter, it is important that we know what exactly the molar mass of an element is.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
